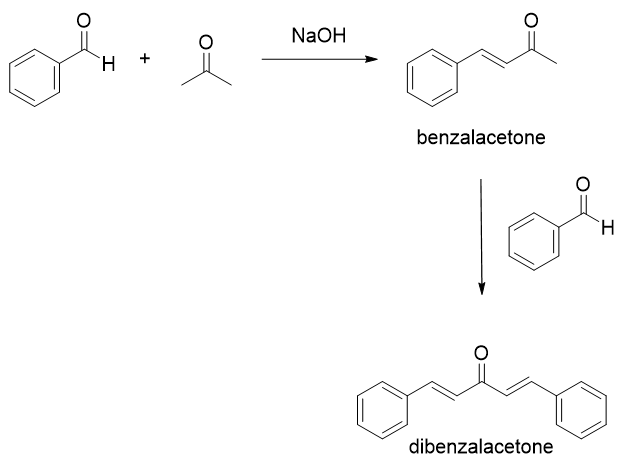
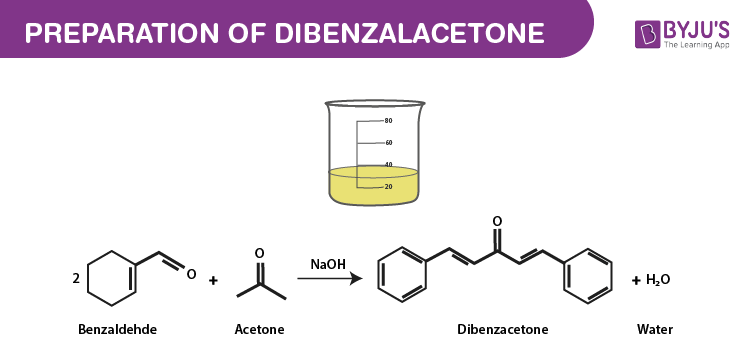
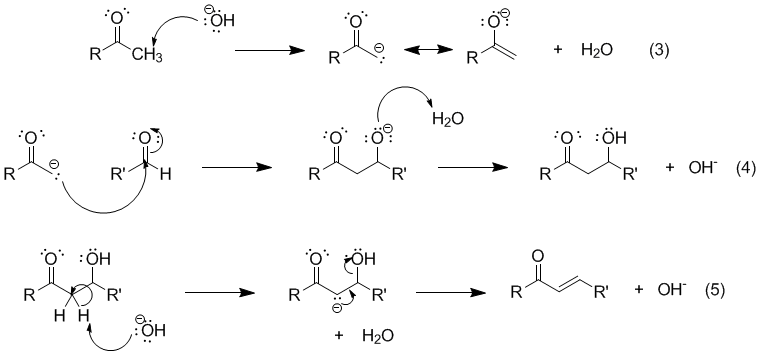
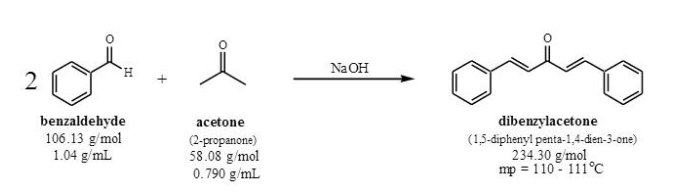
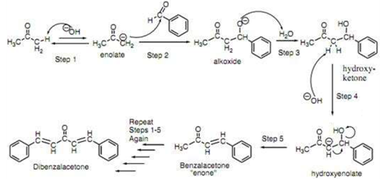
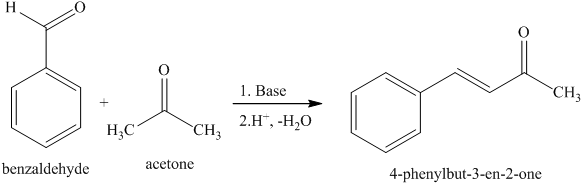
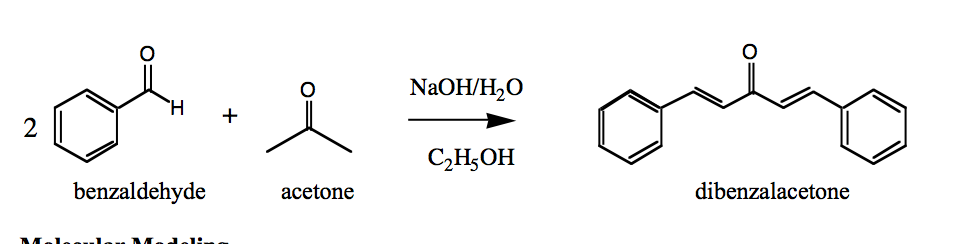
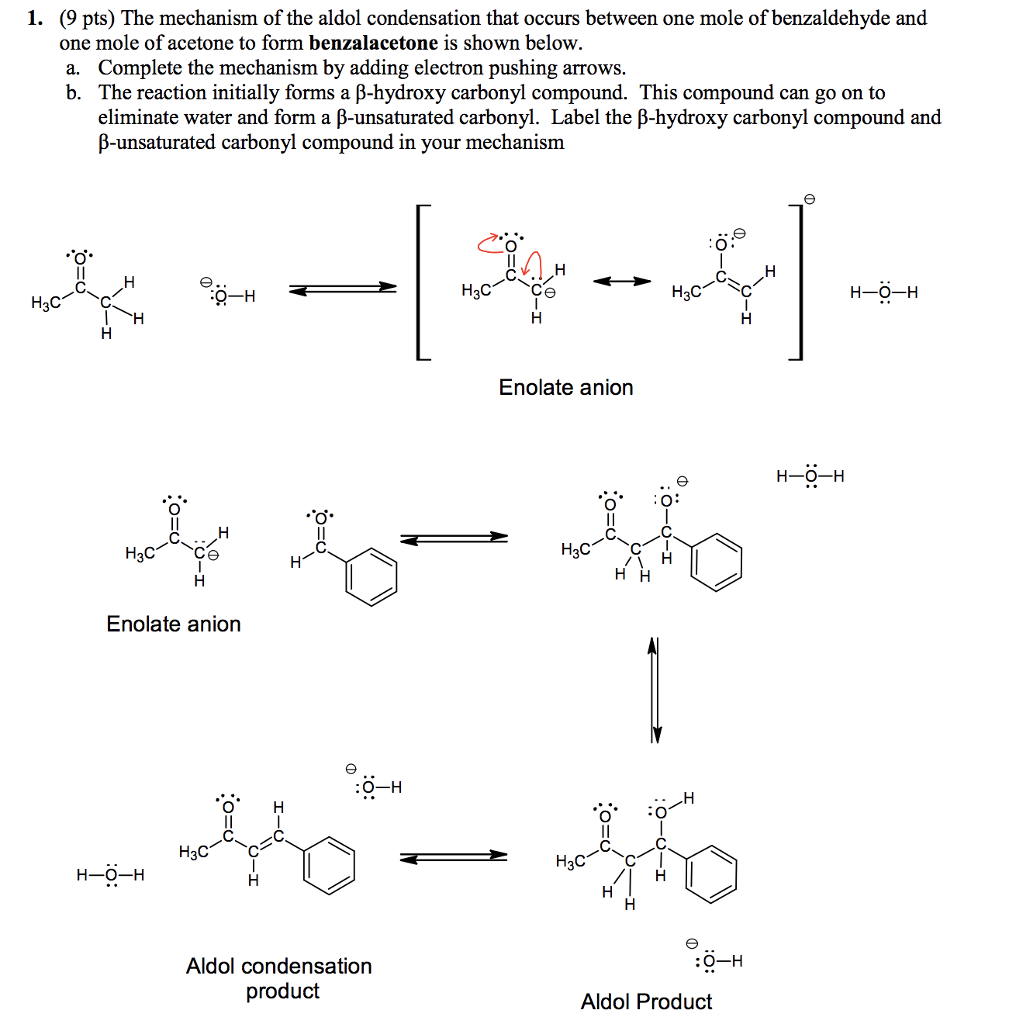
Write out all the steps in the mechanism for the base catalyzed condensation of acetone with 2 mol of benzaldehyde. | Homework.Study.com

Synthesis of 4-Hydroxy-4-(4-nitrophenyl)butan-2-one using p-Nitro Benzaldehyde and Acetone in Aqueous Micellar Media using L-Proline | Semantic Scholar
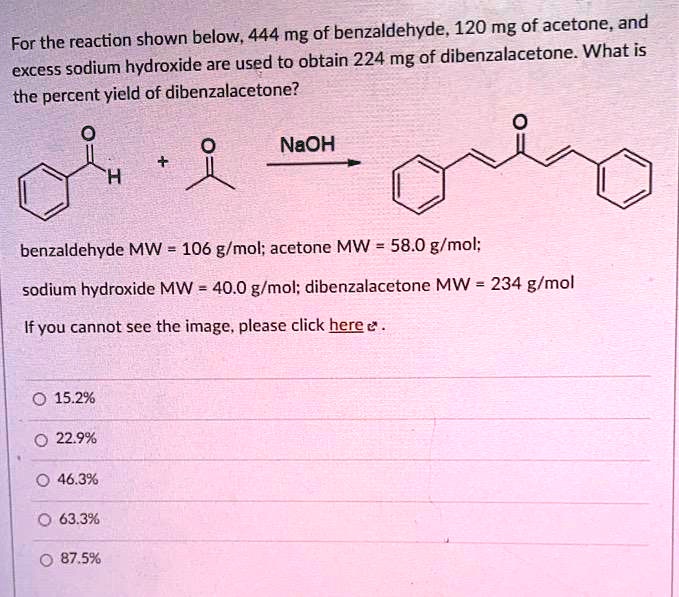
SOLVED: shown below, 444 mg of benzaldehyde; 120 mg of acetone and For the reaction excess sodium hydroxide are used to obtain 224 mg of dibenzalacetone What is the percent yield of

The product formed by aldol condensation between benzaldehyde and acetone is benzalacetone. Its structure is:

The condensation reaction between one equivalent of acetone and two equivalents of benzaldehyde in presence of dilute alkali leads to the formation of:
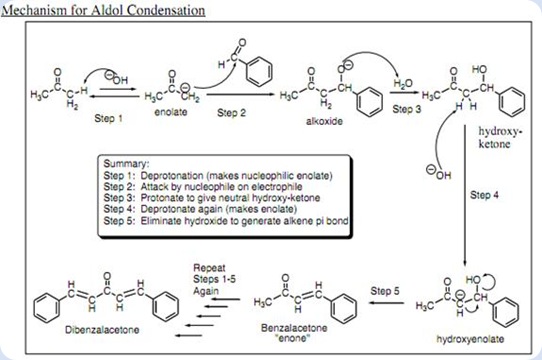
If given this balanced equation: 2C_7H_6O + C_3H_6O -> C_17H_14O +2H_2O Why is the acetone deprotonated and not the benzaldehyde? | Socratic

Write a reaction that would take place during the synthesis of dibenzalacetone if the concentration of acetone in the reaction mixture is not carefully controlled. | Homework.Study.com
Scheme 1 Mannich reaction between benzaldehyde 1, aniline 2 and acetone 3. | Download Scientific Diagram