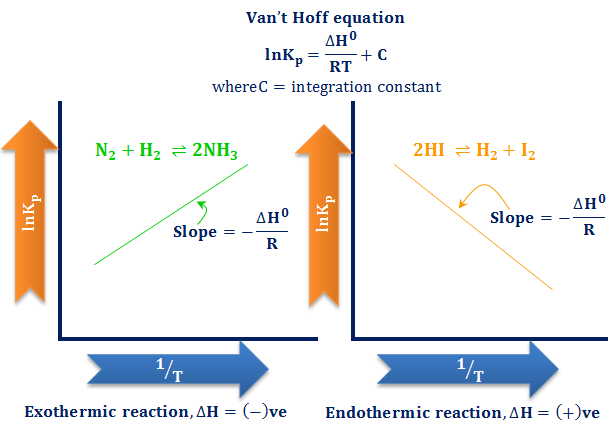
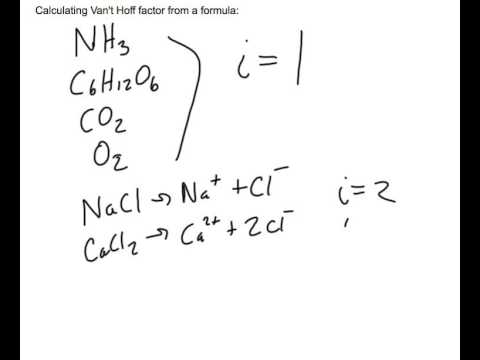
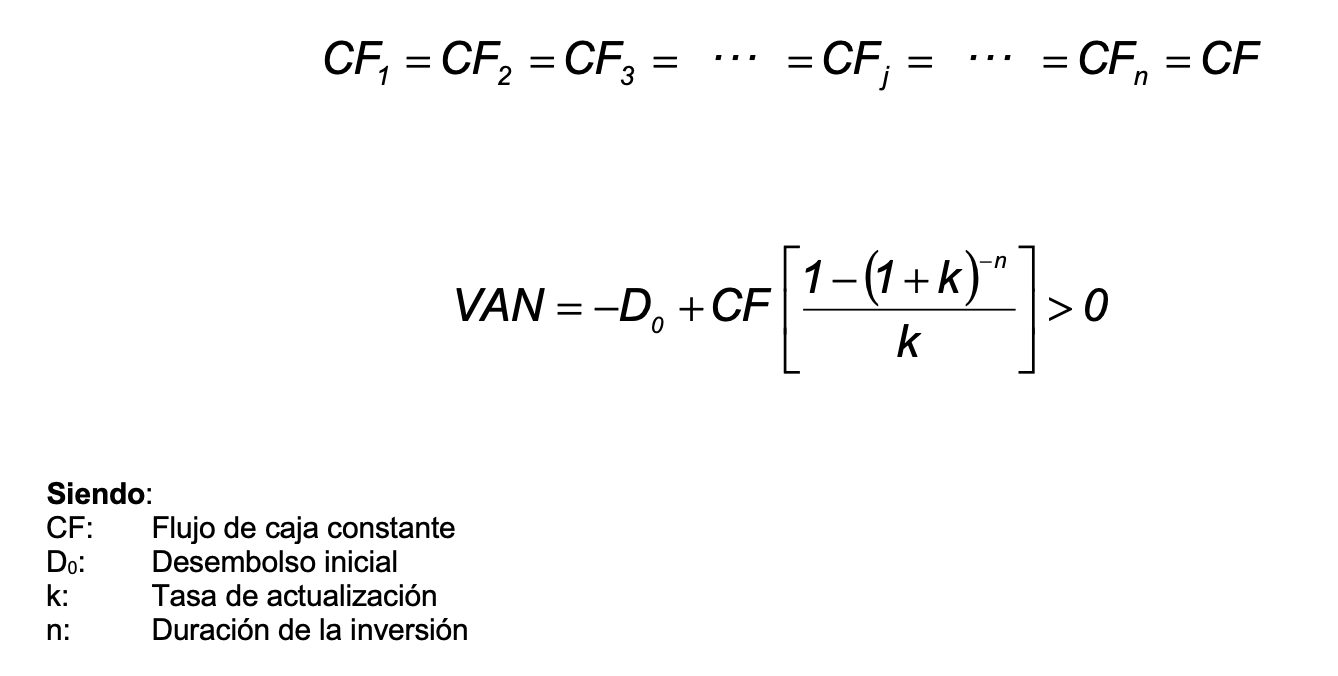
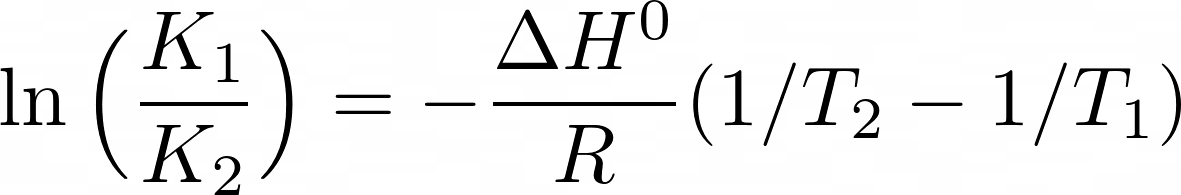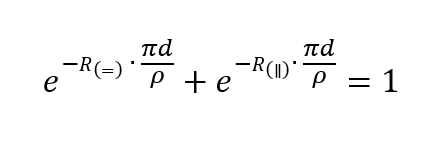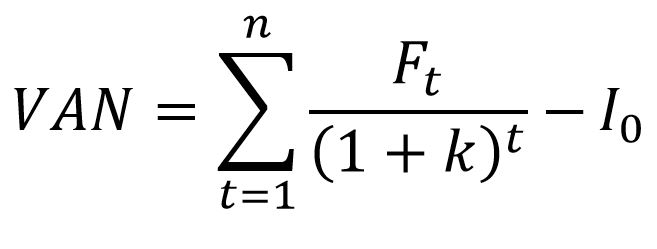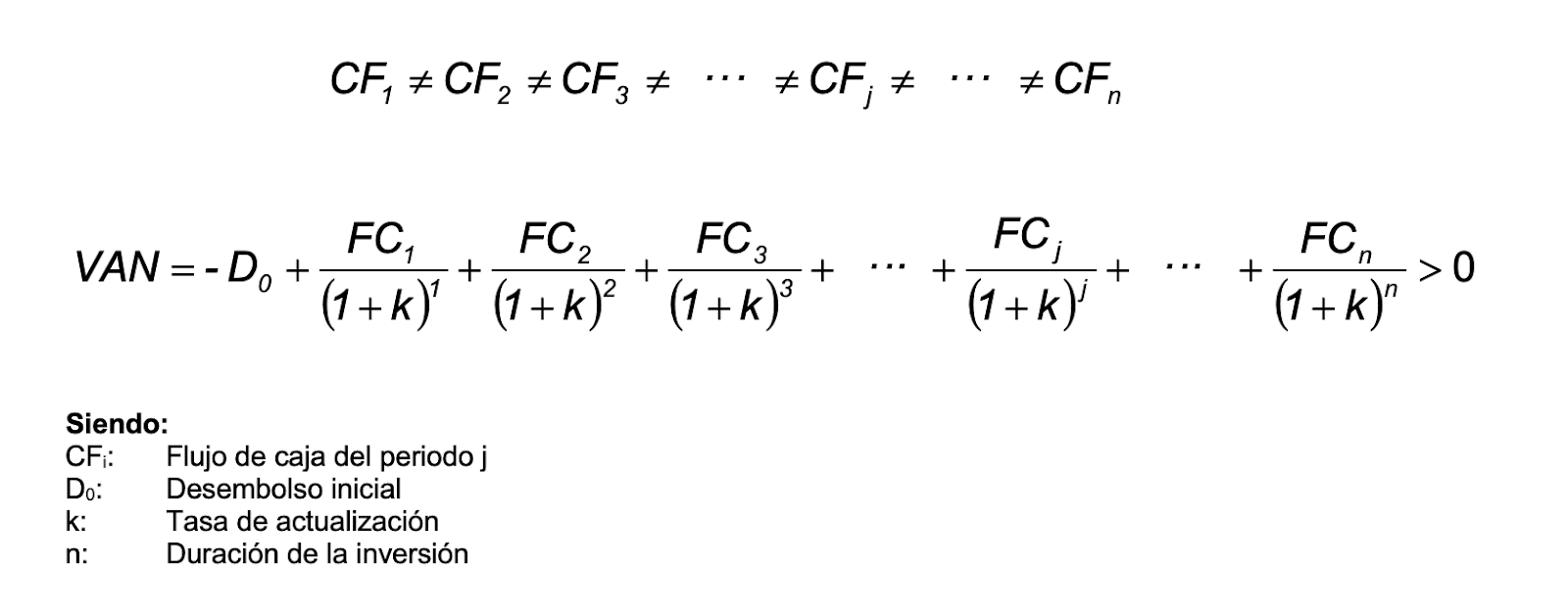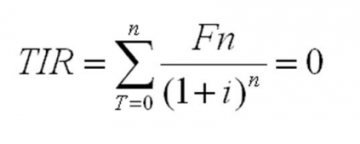SOLVED: An aqueous solution containing 0.250 mole of Q, a strong electrolyte, in 5.00 × 10^2g of water freezes at -2.79^∘C . What is the van't Hoff factor for Q? The molal

Formula 3 - Doohan puts debutants Van Amersfoort first on Day 1 in Valencia | Federation Internationale de l'Automobile

Van Amersfoort Racing to join F3 next season as series confirms all ten teams for 2022-2024 | Formula 1®
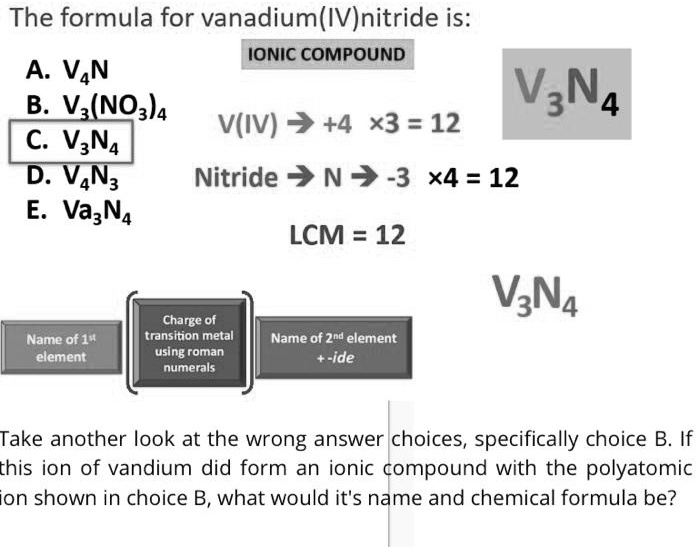
SOLVED: The formula for vanadium(IVInitride is: IONIC COMPOUND A VAN VzN4 B.Vz(NO3)4 V(IV) 7+4 x3 =12 C VzN4 D: VAN3 Nitride 7 N v -3 x4 = 12 E. VazNA LCM =